ISO 14971 Risk Management Consulting
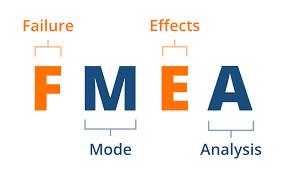
Complete Risk Management Files: plans, hazard analyses (HACCP/HAZOP), DFMEA/PFMEA, benefit-risk analysis, and post-market surveillance integration. Our frameworks support submissions to FDA, EU MDR, Health Canada, and SFDA.
- Comprehensive RMF Development
- DFMEA & PFMEA Engineering
- Post-Market Surveillance Integration


